a) Fe + 2HCl -> FeCl2 + H2
\(m_{HCl}=200 \times 7,3\)%=14,6 (g)
\(\rightarrow n_{HCl}=0,4(mol)\)
\(n_{Fe}=\frac{n_{HCl}}2{}=0,2(mol)\)
\(\rightarrow m_{Fe}=11,2(g)\)
\(m_{Cu}=17,6-11,2=6,4(g)\)
b) \(n_{H_{2}}=n_{Fe}=0,2(mol)\)
\(m_{ddspu}=11,2+200-0,2\times 2=210,8(g)\)
\(n_{FeCl_{2}}=n_{Fe} =0,2(mol)\)
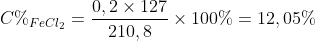
c) Cu + 2H2SO4 -> CuSO4 + SO2 + 2H2O
\(n_{Cu}=n_{SO_{2}}=0,1(mol)\)
\(V_{SO_{2}}=0,1\times22,4=2,24(l)\)